Protein is often considered primarily in terms of “daily intake amount.” However, nutritional research over the past two decades has shown that the biological value of protein depends not only on quantity but also on the quality of protein in the diet. This is particularly significant in the context of shifting dietary patterns, increased consumption of processed foods, and the growing trend toward plant-based protein intake.
According to the Food and Agriculture Organization (FAO, 2013), dietary protein quality reflects a food’s ability to provide sufficient essential amino acids and nitrogen to meet the metabolic needs of the human body. Three fundamental factors determine protein quality: (1) essential amino acid composition, (2) digestibility, and (3) the bioavailability of amino acids after absorption. The recommended protein intake for adults is approximately 0.83 g/kg/day, while older adults require higher intakes to maintain muscle mass and physical function (Matthews et al., 2025).
Protein Quality Assessment Indices
Methods for evaluating protein quality have been developed to compare the nutritional value of different protein sources. The Protein
Digestibility-Corrected Amino Acid Score (PDCAAS) was previously the widely accepted standard; however, the FAO now recommends the Digestible Indispensable Amino Acid Score (DIAAS), as this method assesses amino acid digestibility at the ileal level and calculates digestibility separately for each essential amino acid.
Nevertheless, chemical indices mainly reflect the compositional characteristics of foods and do not fully describe the metabolic activity of amino acids within the body. Therefore, research approaches using stable isotope techniques have been applied to evaluate amino acid bioavailability and their postprandial appearance in systemic circulation.
Differences Between Protein Sources
Recent nutritional studies indicate that protein quality may differ between animal and plant sources. Animal proteins generally provide a well-balanced essential amino acid profile and exhibit high digestibility, whereas some plant proteins may be limited in certain essential amino acids such as lysine or methionine (Matthews et al., 2025).
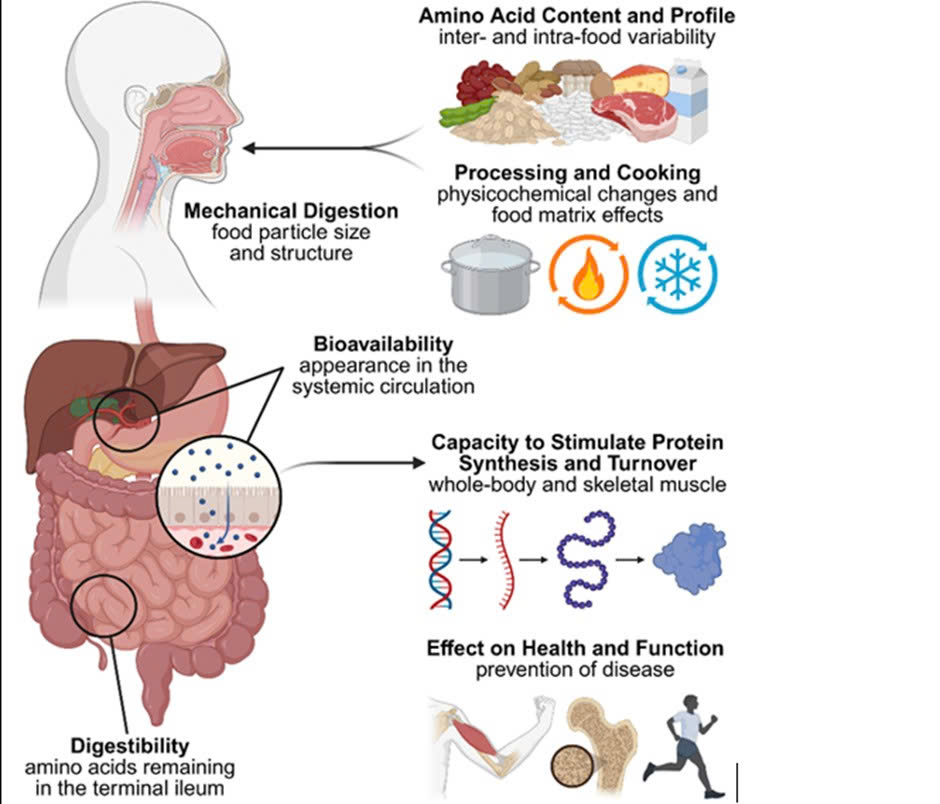
Figure 1. Factors Influencing Dietary Protein Quality
Protein quality is influenced by essential amino acid composition and digestibility, and it may also vary depending on food processing, cooking methods, and mastication–digestive processes. After absorption, amino acids are utilized by the body for protein synthesis and metabolism, with efficiency differing according to dietary patterns and physiological status.
However, diversified and complementary dietary patterns can significantly improve the nutritional value of protein, such as combining proteins from cereals and legumes. In addition, protein isolates derived from soy, potato, or plant-based meat alternatives can achieve protein quality comparable to certain animal protein sources in terms of amino acid composition and digestibility (Matthews et al., 2025).
The Role of Food Processing
Matthews et al. (2025) observed that cooking methods can significantly influence the nutritional value of protein.
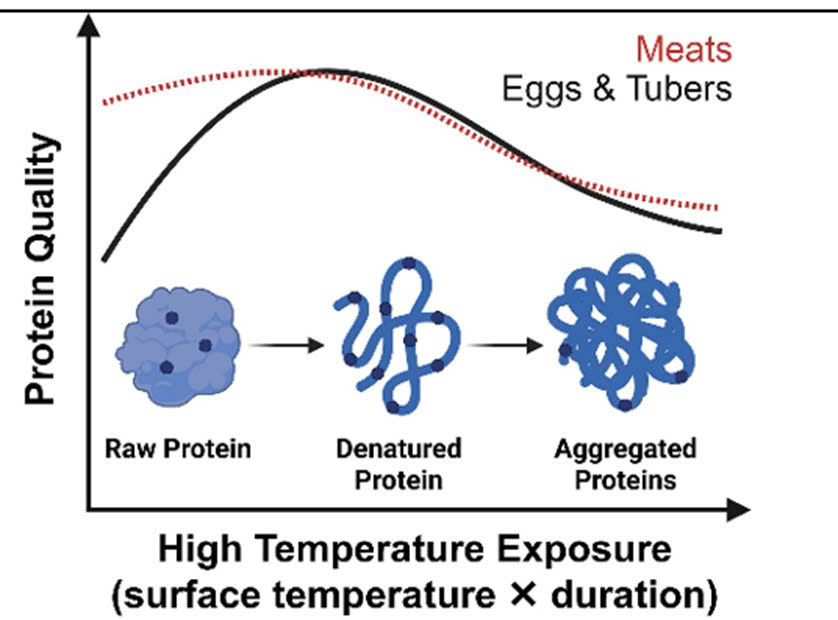
Figure 2. The Effect of Cooking on Protein Quality
The solid line in the graph illustrates the trend in protein quality changes during cooking in certain foods, such as eggs and root vegetables.
The red dashed line represents the case of some meats (e.g., beef), where protein digestibility changes very little between the raw state and moderate cooking.
Moderate cooking generally enhances protein digestibility due to protein denaturation and the reduction of anti-nutritional factors. In contrast, prolonged exposure to high temperatures or intensive heat processing may reduce available lysine through the Maillard reaction, thereby lowering overall protein quality. This has practical implications for diets heavily reliant on processed foods, which are increasingly common in modern urban lifestyles.
Protein and Muscle Protein Synthesis
Protein provides amino acids necessary for muscle protein synthesis and the maintenance of whole-body protein balance. The capacity of a protein source to stimulate muscle protein synthesis largely depends on its essential amino acid (EAA) content, particularly leucine (Matthews et al., 2025). Leucine plays a critical role in activating the mechanistic target of rapamycin (mTOR) signaling pathway, a central regulator of muscle protein synthesis. Physiological studies indicate that proteins rich in essential amino acids elicit a stronger muscle protein synthetic response compared with lower-quality proteins, even when total protein intake is equivalent.
Conclusion
Contemporary nutrition research demonstrates that protein quality depends not only on the total amount consumed but also on essential amino acid composition, digestibility, and amino acid bioavailability. These factors determine the extent to which the human body can utilize dietary protein for growth, maintenance of muscle mass, and metabolic health.
Current recommendations regarding protein quantity and quality are primarily based on guidelines from the World Health Organization (WHO), the Food and Agriculture Organization (FAO), and other international nutrition authorities, which emphasize the importance of essential amino acids, digestibility, and bioavailability in food proteins. A proper understanding of protein quality supports the development of balanced dietary patterns aligned with modern lifestyles and contributes to improved public health outcomes.
References
1. Matthews JJ, Arentson-Lantz EJ, Moughan PJ, Wolfe RR, Ferrando AA, Church DD. Understanding dietary protein quality: Digestible indispensable amino acid scores and beyond. The Journal of Nutrition. 2025;155:3152–3167.
DOI: https://doi.org/10.1016/j.tjnut.2025.07.005
2. FAO. Dietary protein quality evaluation in human nutrition. Rome: Food and Agriculture Organization of the United Nations; 2013.
Available from: https://openknowledge.fao.org/handle/20.500.14283/i3124e